ZECUITY: New Migraine Treatment Coming Soon…
An old and yet brand new migraine treatment is about to be released – the ZECUITY migraine iontophoretic transdermal system (TDS)!
ZECUITY has gone through a lot of changes. It was formerly called the Zelrix Migraine Patch, and it was developed by NuPaths. It was approved this past January. Earlier this year, NuPathe was acquired by Teva Pharmaceutical Industries, a company based in Israel.
Through all this change, ZECUITY has survived. Teva has talked about launching new migraine treatment on the market this year, although the rumour now is that it may not be available to consumers until 2015.
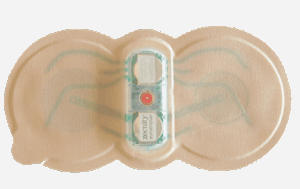
ZECUITY is new and old because the medication is sumatriptan, an early migraine medication. But the delivery method is all new, and for some patients that makes the difference between a drug that doesn’t work and a drug that does.
 |
Dr. Deborah E. Tepper from the Cleveland Clinic describes it this way:
The sumatriptan patch consists of a single-use patch that comes in a foil packet. After tabs are pulled to expose a small well of sumatriptan and another well of salt solution, these areas are each rubbed to release the two compounds. The patch is placed on the upper arm or thigh, in an area where there are no tattoos or skin irritation. A tiny battery embedded in the patch is then turned on by pushing a button. A red light comes on, indicating the patch is activated.
[Source]
When the medicine has all been delivered into your system, the light goes off. Easy as that.
The beauty of this approach is that it bypasses the stomach and gets into your system a lot faster, which may make all the difference. It also means no needles, for those who have been using, or have tried to use, injections.
If the patch is successful, we will likely see more migraine meds coming out with a similar delivery system.
If you’re in the USA and would like to get news directly from the source when ZECUITY is available, you can sign up right here.

MigraineMisfits
15 November 2014 @ 11:55 am
New delivery system for #sumatriptan coming on the market soon. ~Nikki http://t.co/OY1PIG3JXg
nurse_lele
15 November 2014 @ 12:26 pm
RT @MigraineMisfits: New delivery system for #sumatriptan coming on the market soon. ~Nikki http://t.co/OY1PIG3JXg